
Material Identification in Radiographic Images of Dynamically Formed Metal-Explosive Mixtures by Tuning the X-Ray Source Spectrum Using Multiple Anodes

Project # 23-061 | Year 3 of 3
Brandon La Lonea, Dan Sorensona, Ruben Valenciaa, Tom Hartsfieldc, Stuart Bakerb, Stuart Millerb
aSpecial Technologies Lab (STL), bLos Alamos Operations (LAO), cNational Lab: Sandia National Laboratories
This work was done by Mission Support and Test Services, LLC, under Contract No. DE-NA0003624 with the U.S. Department of Energy, the NNSA Office of Defense Programs, and supported by the Site-Directed Research and Development Program. DOE/NV/03624–1931.
Abstract
The goal of this project is to quantify the amount of a particular element in a mixed ejecta field on a dynamic experiment. We use two-angle flash radiography for the material identification where the radiographic sources have different anodes that alter their x-ray spectrum. In one x-ray diode we use a tantalum anode and in the other we use a gold anode. The target element in the ejecta field is zinc. The characteristic L-lines of tantalum are just below the zinc absorption k-edge and the L-lines in gold are just above the k-edge. Therefore, zinc more strongly absorbs the gold x-rays than the tantalum x-rays, allowing us to identify zinc in a background of other elements. In the first year, we demonstrated this effect statically in preparation for dynamic experiments in the second year.
Background
When an explosive driven shock wave reflects from the free surface of a metal, the metal can particulate into ejecta particles. The sources of the ejecta are features in the metal such as machine marks, divots, or joints between materials. In some cases, it may be possible for the high explosive (HE) gases to breech the metal such that the ejecta is a mixture of metal particles and HE products. These mixed ejecta fields are of interest to this work. There have been efforts at Los Alamos National Laboratory, the Atomic Weapon Establishment, and the Nevada National Security Sites to study mixed ejecta fields and quantify the amount of HE products present (if any). Approaches taken previously were to study with imaging, x-ray radiography, and laser induced breakdown spectroscopy (LIBS). The prior work is unpublished but to date has had limited success in quantifying the amount of HE products in the ejecta field. Therefore, another approach is needed—one that can quantify the individual constituents in the ejecta field.
Technical Approach
The approach taken in this project is to quantify the amount of HE products in a mixed ejecta field using two-color (and two-angle) flash radiography where the x-ray source spectra are tuned by using different anode materials. In a typical x-ray diode, the x-rays are created by the collision of high-energy electrons with an anode. These collisions generate both Bremsstrahlung and characteristic x-rays. The Bremsstrahlung x-ray spectrum is very broad and featureless and extends in energy from essentially 0 keV up to the end point energy determined by the impinging electron energy, which in turn is determined by the voltage of the x-ray source. For example, our x-ray source has a peak voltage of 380 kV, so the spectrum has an end point energy of 380 keV. The characteristic x-rays are a set of narrow lines at discreet energies that are specific to the anode material. They result from the target atom’s inner shell electrons being ejected, then as higher orbital electrons drop in energy to fill the vacancy, the atoms emit an x-ray at a specific energy given by the transition energy. Because they depend on the interatomic transition energies of the anode, the characteristic x-rays are unique to each anode material.
The x-ray absorption spectrum of the ejecta also has discrete features due to the atomic composition. These are called x-ray absorption edges and occur where the x-ray energy is equal to the energy required to eject an inner shell electron from the atom. At x-ray energies just above the edge, the absorption cross section is much higher than just below the edge. For the k-edge the absorption cross section is typically 5–10x higher above the edge energy than below it.
In this project we take advantage of both of the above effects to identify a specific atomic species in the ejecta field. We use two different x-ray sources; one has a gold anode, and one has a tantalum anode to take simultaneous flash x-ray images of a scene with the goal of detecting the presence of zinc. The zinc absorption k-edge occurs at an energy of 9.66 keV, and the attenuation coefficient changes discontinuously from a value of 35 cm2/g below the edge to 254 cm2/g above the edge; an increase of ~ 7x. The characteristic L-lines of Ta are at 8.09, 8.15, 9.34, 9.65, and 10.90 keV, so that all but one are below the zinc k-edge. The characteristic L-lines of Au are at 9.63, 9.71, 11.44, 11.58, and 13.38 keV, so that all but one are above the zinc k-edge. The net result is that zinc more strongly absorbs the gold x-rays than it does the tantalum x-rays, whereas in every other element the absorption is similar (actually the gold x-rays are slightly more penetrating than the tantalum x-rays for all elements besides zinc). We manufactured gold and tantalum anodes for the Special Technologies Laboratory (STL) super saver x-ray sources. These are 380 kV flash x-ray sources with a rod-pinch diode design and a nominally ~30 ns pulse width. The differential absorption effect was assessed on zinc and plastic step wedge foils. We are in the process of transitioning to a dynamic experiment at the STL boombox and are also developing analysis software.
Results and Technical Accomplishments
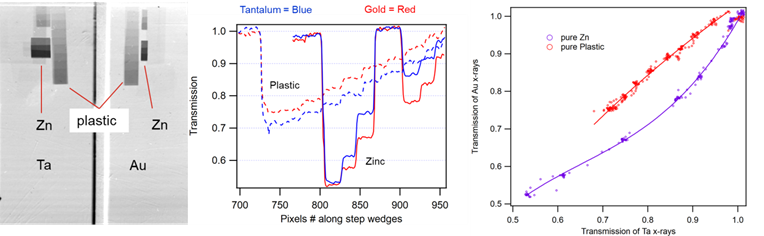
The attached figure (left) shows an example radiographic image of zinc and plastic step wedges taken with the gold and tantalum x-ray sources. Transmission line outs (middle) show how there is less transmission of gold x-rays through the zinc steps wedges compared with the tantalum x-rays, especially on the first few steps. The zinc foil thicknesses were 2.5, 5.0, 7.5, 15, 30 and 45 microns. The transmission values through the plastic steps were higher for the gold x-rays than the tantalum x-rays, which is the opposite trend from the zinc. The plastic thicknesses were 100, 200, 300, 400, 500, 750, 1000, 1250, and 1500 microns. A plot of gold x-ray transmission versus tantalum x-ray transmission (right) for the same objects (either zinc or plastic) shows how one can distinguish between zinc and plastic using the two x-ray sources. If the transmission values for a particular object lie along the upper curve than it is plastic, if the values lie along the lower curve than it is zinc. If the transmission values lie between the two curves than it is a mixture of zinc and plastic with a ratio that we will be able to estimate once our analysis routines are complete.
Conclusions and Path Forward
We have developed and evaluated a method for determining the type of material present using x-ray radiography with two different x-ray sources. By using the characteristic x-rays from gold and tantalum anodes we can distinguish zinc metal from plastics, and in general zinc atoms from any other atomic species present. In upcoming dynamic experiments, we will use an explosive doped with zinc oxide to generate an ejecta plume (of likely tin metal). Using this approach, we will be able to locate and quantify the amount of zinc in the ejecta plume, hence determine how much of the high explosive products are present.
Publications
- Title: Material identification in radiographic images of mixtures by tuning the x-ray source spectrum using multiple anodes
Journal / Conference: JOWOG 32 HDT
Year: 2023
Author(s): B. La Lone, D. Sorenson, B. Valencia, T. Hartsfield, J. Goett, S. Baker, D. Morgan, S. Miller, N. Maserang
