
New Methods to Study the Kinetics of Phase Transformation in Shocked Cerium Metal

Project #: 22-050 | Year 2 of 2
Gerald Stevens,a Brandon La Lone,a William Turley,a Thomas Hartsfield,b Kevin Baldwin,b Matthew Beasonb
aMission Support and Test Services, Special Technologies Laboratory (STL); bLos Alamos National Laboratory (LANL)
Executive Summary
We studied shockwave induced liquid-to-solid phase transformations in the lanthanide metal cerium. The precise temperature (T) versus pressure (P) location of a phase boundary and the rate of phase transformation are difficult properties to measure, and we used recently developed optical reflectometry and radiance techniques to probe the cerium phase diagram. For the first time, we were able to detect refreezing of shocked cerium in dynamic experiments using thin (2 μm) film samples. We showed that for films shock-melted and held at constant pressure, the kinetics of the refreezing transformation were faster than about 10 ns.
Description
We teamed with the LANL shock physics and material science community to develop new methods to investigate equation of state properties of cerium and the kinetics of shock-induced phase transformations. We have previously developed a compact diagnostic tool to simultaneously measure velocity (which infers stress), radiance and reflectance (which give us temperature) of a shocked sample. Several years ago, we used this tool to study cerium in a unique set of experiments where the sample was shocked, held at pressure and then released to low pressure. During the hold cycle, we observed, as expected, very little change in the T and P of the target material and thus were able to determine a T-P Hugoniot point from each experiment between 8.4 and 23.5 GPa.
During this early study, we observed unexpected changes in the sample reflectivity during the hold cycle of about 600 ns that may have indicated a gradual phase transformation was occurring. However, based on recently published work, we were concerned that our results may have been affected by sample surface contamination. We subsequently began to develop methods of handling and preparation to ensure our surface reflectivity measurements were from pure cerium metal. We proceeded with two approaches: 1) use conventional machining methods to quickly “diamond turn” the cerium sample surface in argon gas, removing surface contamination while inhibiting oxidation, and then promptly gluing to a LiF window and loading into the evacuated experiment chamber, and 2) sputter a thin coating of cerium onto a LiF window and then overcoat this film with a thin aluminum layer to protect the cerium sample from reacting with the surrounding atmosphere.
In our first dynamic experiment, we used a 6 mm thick LiF window coated with a 2 μm film of cerium overcoated with a 0.5 μm aluminum film protecting the cerium. For the results shown, a thin film sample was impacted with an aluminum flyer plate and the velocity, radiance, and reflectance from the cerium film was measured through the LiF window. This is a novel new measurement technique in which the cerium film is shock heated and melted, and while the pressure is held steady, it rapidly cools by conduction (red trace) into the surrounding cooler aluminum and LiF materials. As the sample cools and the radiance rapidly drops, a hesitation in the rate of cooling is seen where the sample resolidifies. The brief pause in cooling as the metal re-freezes suggests that it occurs in 10s of ns. In these experiments, we were able to shock heat into the liquid state at 23 GPa and then observe the sample resolidification while cooling across the liquid-solid phase boundary.
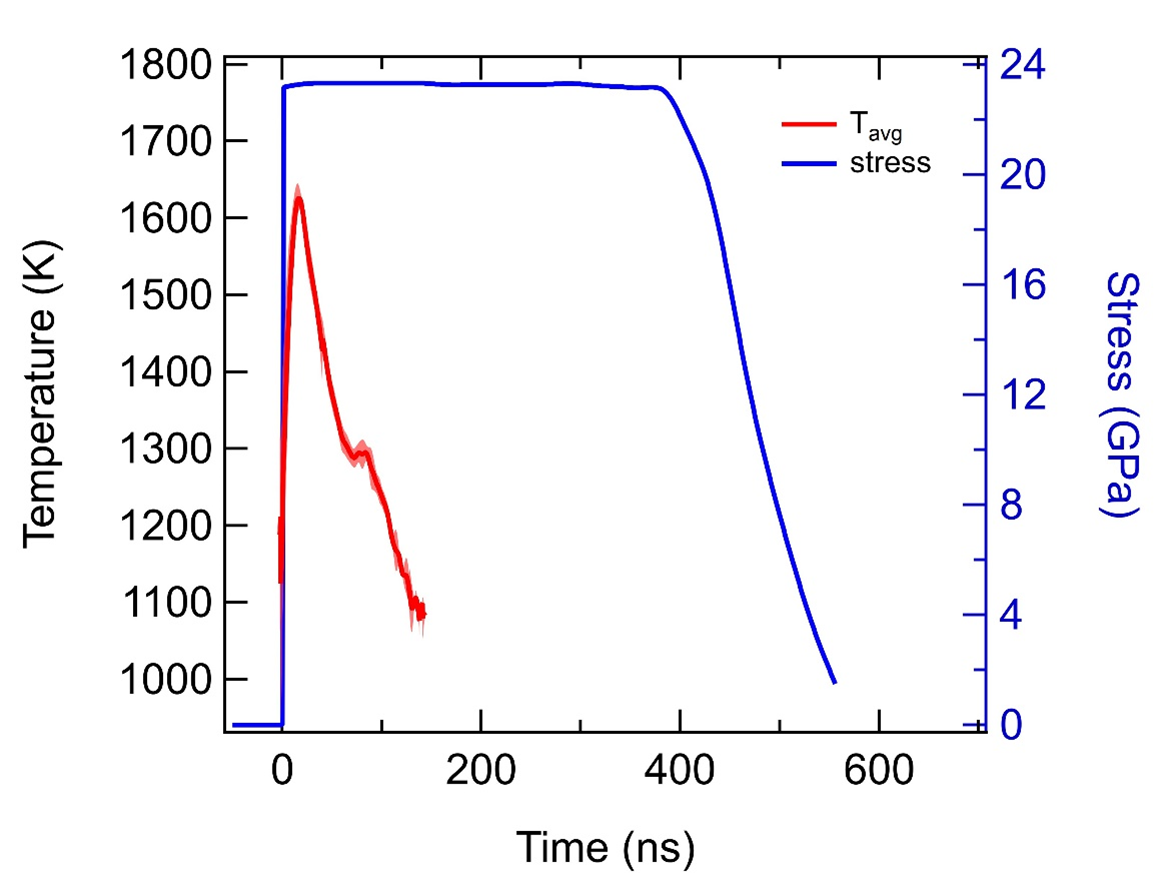
Conclusion
In collaboration with LANL, we have developed methods to prepare the surface of cerium metal to enable dynamic reflectivity and radiance measurements with pristine target samples. We dynamically tested samples prepared with these new methods using the powder gun launcher at STL. For the first time, we demonstrated a new experimental loading technique (isobaric film cooling) that allowed us to observe the refreezing of shock melted cerium. Our results are consistent with the extension of the previously reported diamond anvil cell (DAC) solid-liquid phase boundary and represent a breakthrough methodology in the mapping of phase boundaries at high pressures and temperatures. The transitions we observed took tens of nanoseconds to complete.
Mission Benefit
The isobaric cooling technique is a breakthrough for dynamic mapping of material phase boundaries and we expect a significant scientific contribution resulting from the application of this method to cerium and other materials. A number of benefits from this effort are being realized in the NA-10 Stockpile Stewardship program. Our method for cerium preparation is being applied to dynamic experiments in support of the Lawrence Livermore National Laboratory Shallow Bubble Collapse project sponsored by DOE Primary Assessment Technology. Also, samples have been prepared and used in collaborative experiments with LANL to test a novel diagnostic tool to detect trace quantities of shockwave generated atomic cerium vapor. Finally, the thin film isobaric cooling method is being adapted for future support of LANL Dynamic Material Properties studies.
Publications, Technology Abstracts, Presentations/Posters
Hixson, R. S., B. M. La Lone, M. D. Staska, G. D. Stevens, W. D. Turley, L. R. Veeser. 2021. “Temperature Measurements in Cerium Shocked from 8.4 to 23.5 GPa.” J Appl Phys.129, 155106. https://doi.org/10.1063/5.0043096.
Hartsfield, T. M., B. M. La Lone, G. D. Stevens, M.T. Beason, J. K. Baldwin, W. D. Turley. Manuscript in preparation. “Dynamic Resolidification of Shock-Compressed Cerium Measured by Isobaric Thin Film Cooling.”
References
Akashev, L. A., N. A. Popov, V. G. Shevchenko. 2018. “Optical Properties and Electronic Characteristics of Cerium in the Condensed State.”J Appl Phys.85(4); 624-629.
https://doi.org/10.1007/s10812-018-0696-3.
La Lone, B. M., P. D. Asimow, O. V. Fat’yanov, R. S. Hixson, G. D. Stevens, W. D. Turley, L. R. Veeser. 2019. “High-Pressure Melt Curve of Shock-Compressed Tin Measured Using Pyrometry and Reflectance Techniques.” J Appl Phys.126(22); 225103. https://doi.org/10.1063/1.5132318.
This work was done by Mission Support and Test Services, LLC, under Contract No. DE-NA0003624 with the U.S. Department of Energy. DOE/NV/03624–1641.
Return to Dynamic Experiment Diagnostics
Go to SDRD Annual Report Index
Return to SDRD Homepage
