
Radioactive Noble Gas Detection

Project #: 22-042 | Year 2 of 2
Rusty Trainham,a James Bounds,b Alexandre Kolomenski,b Manuel Manard,a Hans Schuesslerb
aSpecial Tecnhologies Laboratory; bTexas A&M University (TAMU)
Executive Summary
Doppler-free laser spectroscopy of atomic hyperfine structure of xenon has been explored as a means to detect and quantify isotopic and isomeric abundances of noble gases. The technique can potentially offer several orders of magnitude greater sensitivity over standard nuclear coincidence measurements when applied to radioactive noble gases, by virtue of the fact that an entire sample population can be probed, as opposed to the small population of atoms that have undergone radioactive decay during the measurement. We have determined that the optical technique is viable.
Description
Laser spectroscopy permits the identification of isotopes and isomers of atomic species by means of isotope shifts and hyperfine structure. These perturbations of atomic structure arise from interactions of the atomic electrons with the nucleus. The isotope shift is a combination of the reduced mass of the nuclear and electronic attractive force, a volume effect of electronic penetration into the nucleus, and relativistic effects. This shift alters the energy of the electronic atomic state, but it does not create new structure. The hyperfine structure, on the other hand, does create new structure, and it arises from nuclear-spin-dependent magnetic dipole and electric quadrupole moments of the nucleus interacting with the electron. The interaction splits the electronic energy level into multiplets to distinguish not only different isotopes, but also different nuclear isomers of the same isotope. An isomer is an isotope in a nuclear excited state, and the excited nucleus has a different spin than the nuclear ground state, thus it has a different hyperfine structure. Consequently, the hyperfine structure creates a distinctive “fingerprint” of an isotope or isomer. Hyperfine structure exists only for odd half-integer nuclear spin because even integer nuclear spin does not possess magnetic or electric multipole moments. Nevertheless, all of the radioactive species of interest in xenon and krypton are odd half-integer spin. Figure 1 shows a schematic of the current set-up for the laser measurement, as well as a sample of our data. The apparatus comprises a radio frequency discharge cell filled with a few millitorr of xenon, a tunable Ti:sapphire laser, optical components, a chopper wheel, a lock-in amplifier, and photo diodes for measuring absorption and fluorescence. A wave meter and etalon are used to measure the wavelength and bandwidth of the laser. The laser beam is split into two components to illuminate the discharge cell from opposite directions so that the Doppler-free (or sub-Doppler) Lamb dip technique can be exploited.
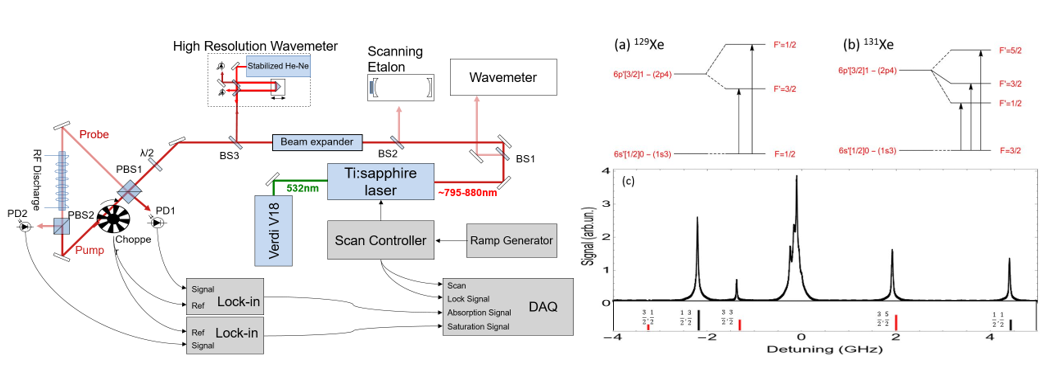
Normally, laser absorption signals from different isotopes fall on top of each other, and the hyperfine structures are interleaved. In a simple absorption or fluorescence experiment, Doppler broadening would wash out the structure we wish to probe. The Lamb dip configuration overcomes the broadening by utilizing counter-propagating laser beams at intensities close to saturation of the probed transition. As the laser is swept through the transition, both beams excite different populations of atoms with opposing velocity components Doppler shifting atoms into resonance. When the laser reaches the natural resonance line width of the transition, the two populations merge and the transition becomes saturated. The two initial state populations merge into one of lesser total population, and a dip (or a peak, depending on experimental configuration) in laser absorption occurs over a frequency width comparable to the natural line width of the transition.
Conclusion
The optical measurement is indeed capable of producing quantitative isotope ratios of natural xenon. The Doppler-free Lamb dip technique can resolve the hyperfine structure of 129Xe and 131Xe, and the isotope shifts of 128Xe, 130Xe, 132Xe, 134Xe and 136Xe. We have not yet performed any measurement of radioactive xenon, but in the near future we are hoping to acquire the medical isotope Xeneisol (133Xe) to confirm our modeling estimates. Measurement of the radioactive isotope will also allow us to compare the sensitivity of our optical technique with traditional nuclear coincidence spectroscopy.
Mission Benefit
Hyperfine spectroscopy can be utilized to measure isotope ratios of noble gas samples, and can potentially increase detection sensitivity by several orders of magnitude over standard nuclear spectroscopic techniques. Smaller volumes of air would need to be processed, and measurement times can be vastly reduced. The technique does not require radiation shielding, so a portable apparatus can fit onto a rolling cart, be loaded into a van, or onto an aircraft, and it can potentially be deployed closer to sites of suspected nuclear proliferation activities, and actionable data can be produced in a matter of minutes.
Publications, Technology Abstracts, Presentations/Posters
Bounds, J., A. Kolomenskii, R. Trainham, M. Manard, H. Schuessler. Forthcoming. “Hyperfine Structure and Isotope Shifts of Xenon Measured for Near-Infrared Transitions with Doppler-Free Saturated Absorption Spectroscopy.” J. Appl. Phys.
This work was done by Mission Support and Test Services, LLC, under Contract No. DE-NA0003624 with the U.S. Department of Energy. DOE/NV/03624–1656.
Return to Enabling Technologies for Autonomous Systems and Sensing
Go to SDRD Annual Report Index
Return to SDRD Homepage
